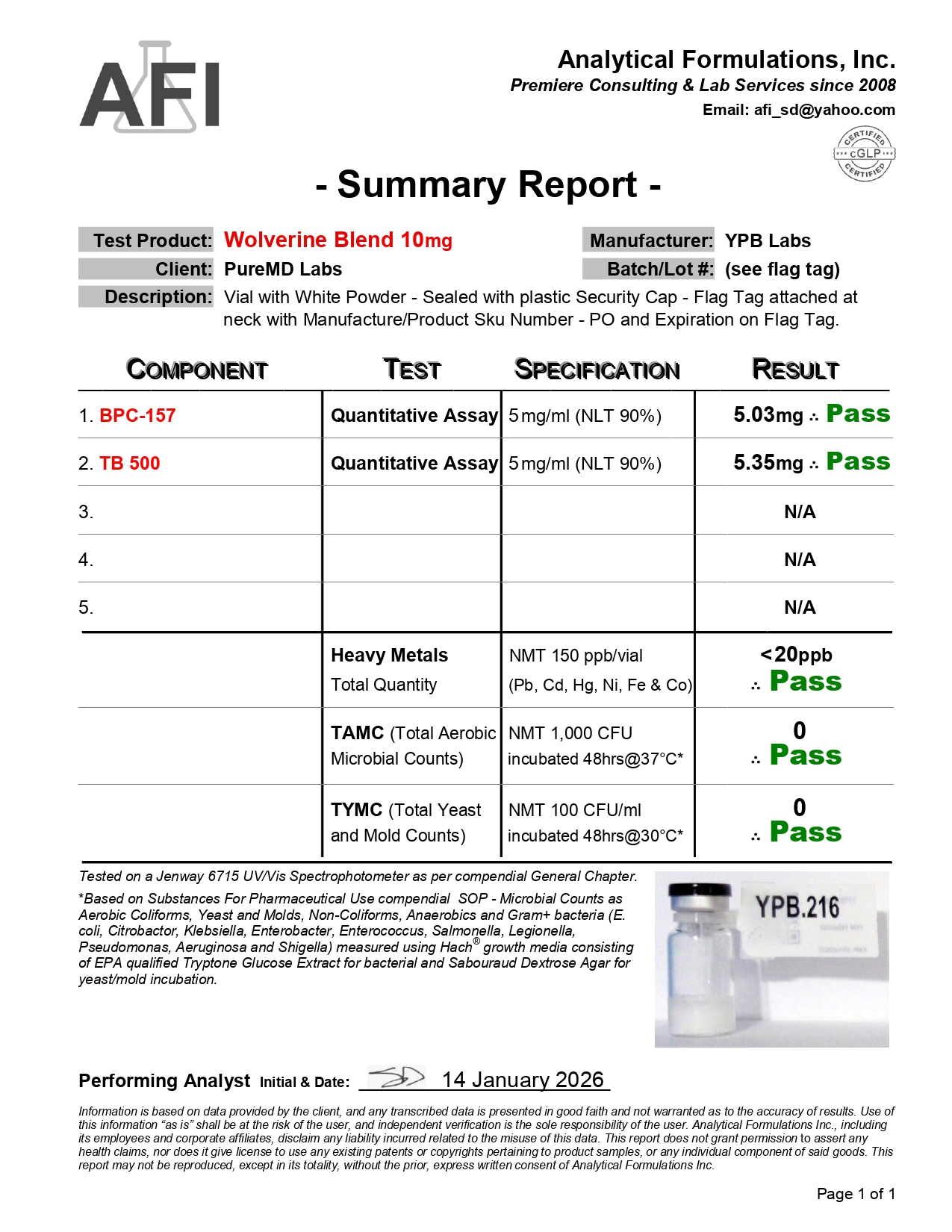
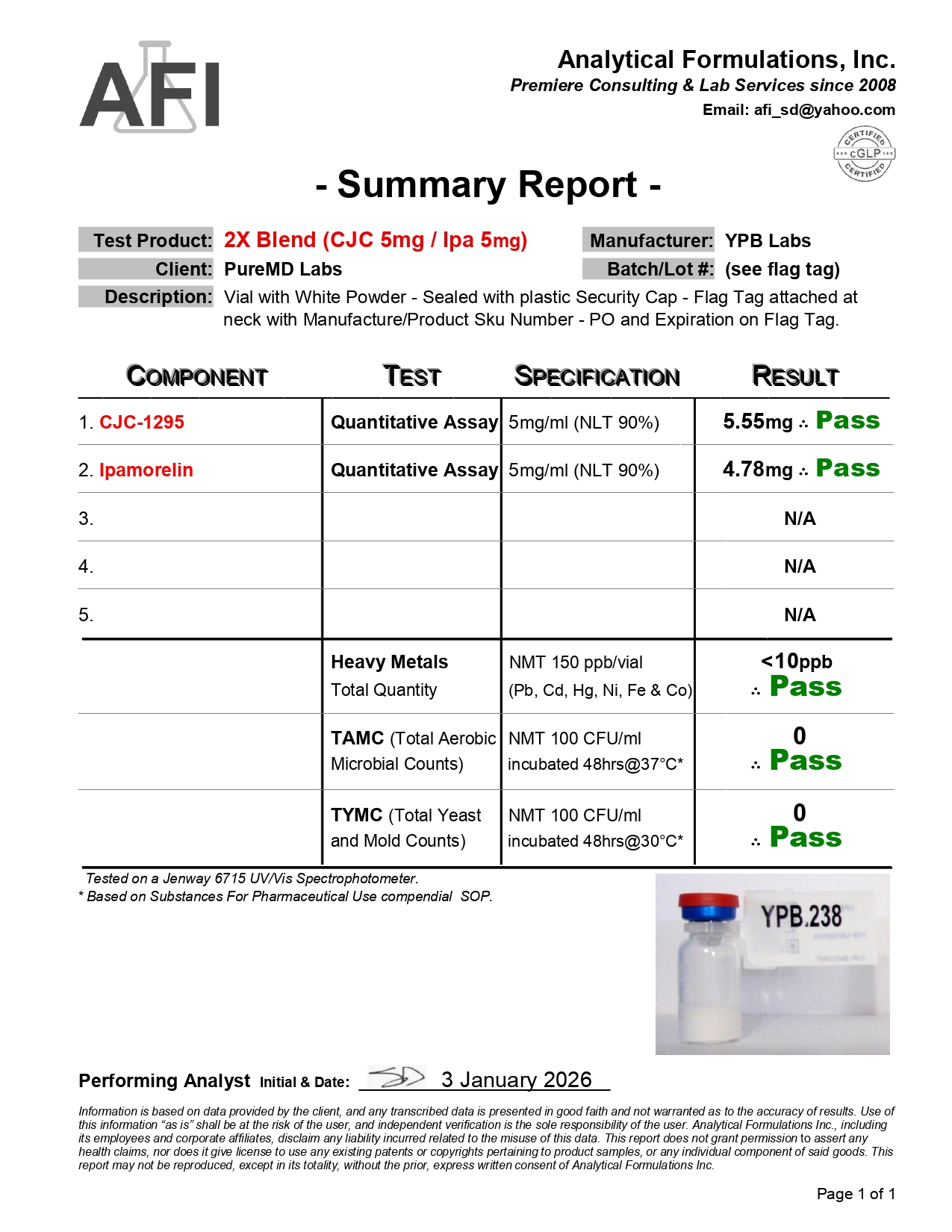
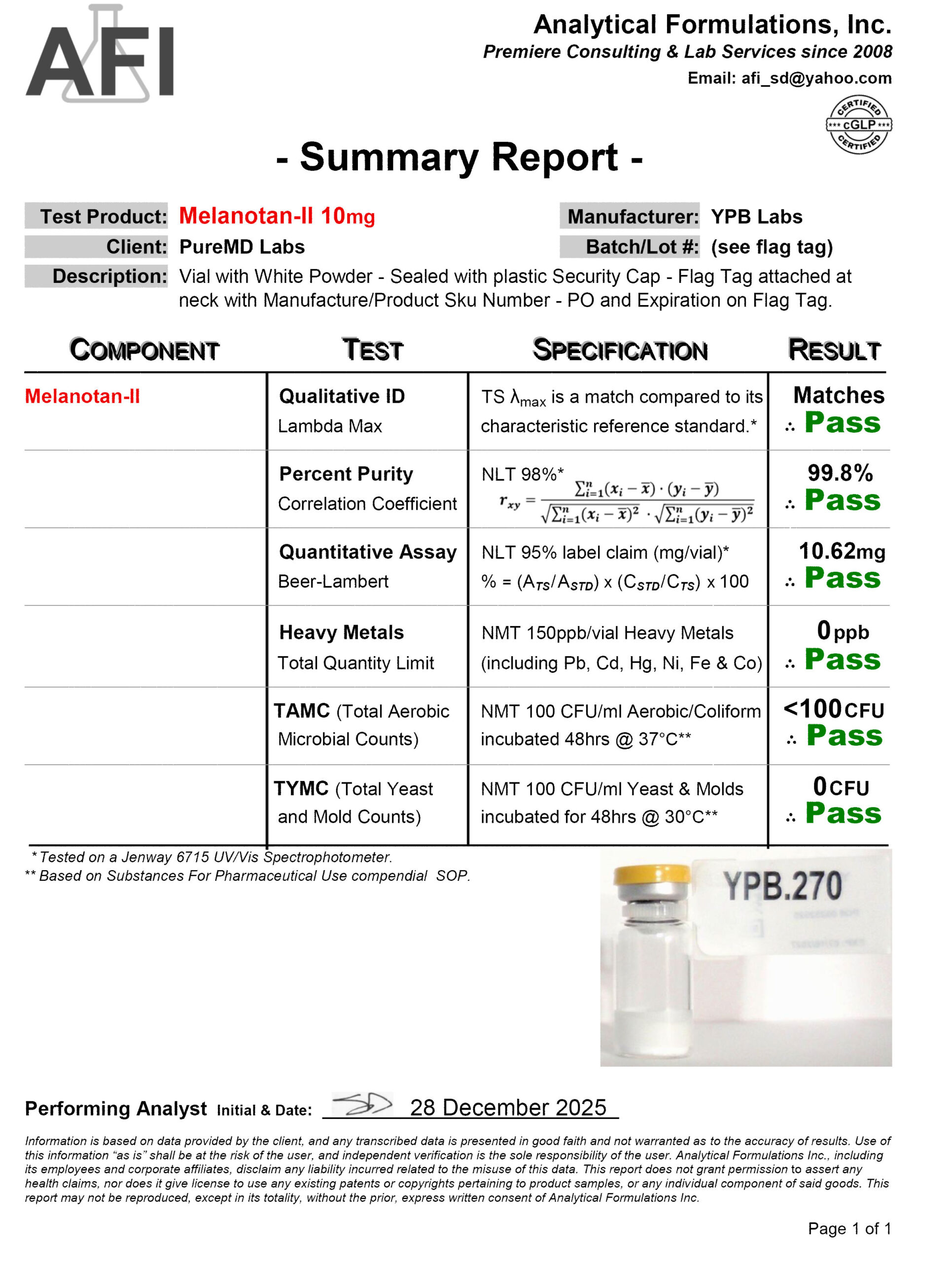
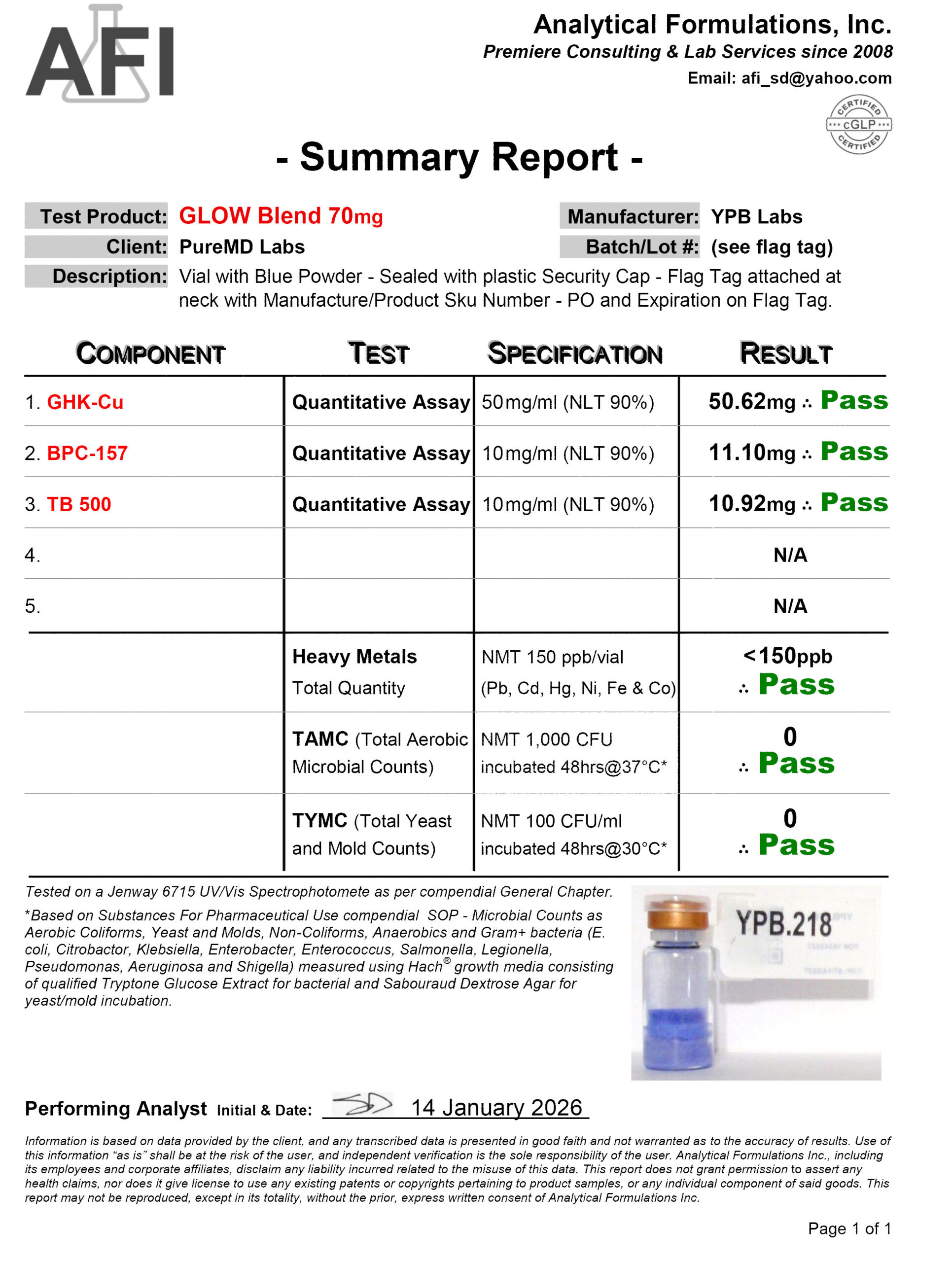
Fast Shipping
Research-Grade Peptides
COAs Available
Fast Shipping
Research-Grade Peptides
COAs Available
Fast Shipping
Research-Grade Peptides
COAs Available
Fast Shipping
Research-Grade Peptides
COAs Available
Fast shipping
Research-Grade Peptides
COAs Available
Fast shipping
Research-Grade Peptides
COAs Available
Fast shipping
Research-Grade Peptides
COAs Available
Fast shipping
Research-Grade Peptides
COAs Available